2D Materials (Graphene)
2D Materials - Graphene Activity Downloadable PDF
Overview:
In this activity students attempt to isolate a one-atom thick layer of graphite (graphene) through manual exfoliation, using a method that won a Nobel prize in 2010.
Materials:
- Nobel Prize Press Release
- Graphite samples
- Clear scotch tape
- A pencil
- White paper
- Digital microscope
- Structure of Graphene image (below)
- Graphene under the microscope image
- Graphene Film (https://vimeo.com/1051348536/0294b34d3d?share=copy)
Preparation:
Watch the Graphene Film prior to students arriving for the activity.
Directions:
- Explain to students that they have the opportunity to participate in an experiment that won a Nobel prize in 2010. Show them the Nobel Prize press release.
- Each student will need a long strip of tape and access to a piece of graphite.
- Give students time to take turns pressing their tape onto the graphite, pressing or rubbing it firmly, and peeling it off.
- Alternatively, some students could sample graphite from a sheet of paper with shading from a pencil to compare (use the side of a pencil to darken an area on paper, then use tape to carefully peel some of the graphite off).
- Students should then fold the tape back onto itself and pull it apart several times (further separating graphite layers). This step is clearly demonstrated in the video.
- Using a digital microscope, view the thin layers of graphite. Layers will likely still appear dark. Single atom layers of graphene are transparent, so not visible using a light microscope and a glass slide. To be able to view a single atom layer, you will need a darker substrate, such as a silicon wafer.
- To view a sample under a microscope using a silicon wafer: Using a small roll of tape, carefully mount the silicon substrate wafer onto a microscope slide using the tweezers. Choose a student to adhere their tape sample onto the silicon wafer. Gently press and rub the sample onto the wafer using a pencil eraser, and then gently remove the tape.
- Students can take turns with this step, as there is only one silicon wafer provided.
- See if you can identify a single layer – graphene. It can be difficult to see – use the demonstration on the Graphene Film (at 3:30) and/or the provided images for reference.
- Play the Graphene Film video for students (optional – could be shown before or after the activity).
Explaining the science:
Graphene is the name of a one atom thick sheet of the material graphite. Graphite
is the material commonly used as pencil “lead”. Researchers. Andre Geim and Konstantin
Novoselov, who successfully exfoliated graphene from graphite, won the Nobel Prize
in Physics in 2010. See the Nobel Prize press release below.
As you will experience through this activity, mechanical exfoliation is a fairly simple
process using simple tools (like tape). It works because of the atomic structure of
graphite is similar to a stack of paper – where sheets peel off but when pulled apart,
the sheet itself is very strong and flexible.
Standard Alignment - coming soon
|
Standard Code |
Standard Text |
How does this activity support this standard? |
This resource was provided by the MSU Science Math Resource Center using resources from the MSU Applied Quantum CORE funded by the Air Force Office of Scientific Research to accompany Graphene Film by MSU School of Film & Photography.
412 Reid Hall • smrc@montana.edu

AlexanderAlUS, https://en.wikipedia.org/wiki/Graphene#/media/File:Graphen.jpg, CC BY-SA 3.0
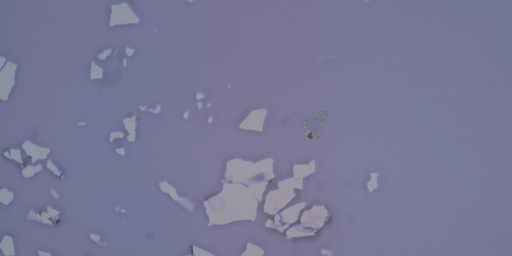
Screenshot from Graphene Film, 3:35
|
Properties |
Graphene |
Graphite |
|
Surface Area |
Extremely high |
Comparatively low |
|
Appearence |
Transparent and colorless |
Opaque and black/gray |
|
Strength |
Strongest material known to exist |
Brittle |
|
Conductivity |
Exceptionally electrically and thermally conductive |
Relatively good conductivity but not as good as graphene |
|
Applications |
Electronics, energy storage, sensors |
Pencils, lubricants, batteries |
Condensed from: https://www.ossila.com/pages/graphene-vs-graphite
